Introduction
Diseases of the ocular surface belong to multifactor diseases. They lead to a decrease in the quality of vision and patient complaints due to changes in the epithelium of the conjunctiva and cornea, and in lacrimal and meibomian glands [1, 2]. Manifestations of dry eye syndrome (DES) may increase with prolonged hypotensive therapy [3], systemic medicamentous therapy [4, 5], and after surgical interventions [6-9].
The incidence of glaucoma according to the World Health Organization (WHO) is from 60 to 105 million people worldwide, and in the next 10 years this number is likely to increase by another 10 million (source: http://www.who.int/ru/) [10]. The prevalence of glaucoma in people over 50 years of age with diseases of the ocular surface exceeds 10% [11-13]. However, the frequency of DES manifestations in patients receiving local hypotensive therapy, according to some data, reaches up to 60% [14-16]. The occurrence of DES against the background of hypotensive therapy leads to a reduction in a patient’s adherence to the therapy [17-19] and, consequently, to the progression of glaucoma [20, 21].
Hence, the condition of the ocular surface in glaucoma patients has a significant impact on the success of local hypotensive therapy, which can lead to the development and/or intensification of DES manifestations [14, 15, 22].
The toxic effect of hypotensive drops is caused by the preservatives in their content. Several classes of preservatives have been developed to date: benzalkonium chloride (BAC), oxidizing preservative Purite® (Allergan Inc., Irvine, CA, USA), detergent Polyquaternum 1 or Polyquad® (Alcon Research, Fort Worth, TX, USA), ionic buffer preservative SofZia® (Novartis AG, Basel, Switzerland), sodium perborate (GenAqua®), and sodium benzoate. Their prolonged use may aggravate symptoms of ocular surface diseases [23-26]. As a result of changes in the composition of a preservative-free carbonic anhydrase inhibitor (CAI) (Dorzolan Solo, Solopharm, Russia) via adding sodium benzoate preservative, patient complaints of burning during instillation became more frequent, which was the reason for conducting the study on changes in the ocular surface condition against the background of using CAI containing sodium benzoate.
The objective was to establish the effect of sodium benzoate on the course of DES and the effectiveness of local hypotensive therapy of glaucoma.
Material and Methods
The study was conducted in the period from September through November of 2020 on the basis of the Municipal Budgetary Healthcare Institution, City Clinical Hospital No. 2, Polyclinic No. 1, and City Glaucoma Office in Chelyabinsk. The data of an analytical, observational case-control study were analyzed. Based on the inclusion and exclusion criteria, 21 patients (41 eyes) were the subject of our study. In all cases, the diagnosis was established sensu the system of differential diagnosis of diseases and confirmed by specific research methods. All patients underwent visual acuity examination, iCare tonometry (Tiolat, Finland), elastic tonometry with Maklakov tonometers (weighing 5, 10 and 15 g), computerized perimetry (CP) (Octopus-600, Haag-Streit Diagnostics, Switzerland), optical coherence tomography (OCT) of the macular zone and optic disc (Spectralis OCT, Heidelberg Engineering, Germany).
At the onset of the study and a month after the change of treatment to CAI containing sodium benzoate, patients were examined for the condition of their ocular surface: tear break-up time (TBUT) sensu Norn, Schirmer test, vital staining with lissamine green, and a survey based on the Ocular Surface Disease Index (OSDI) questionnaire.
The inclusion criteria: city of Chelyabinsk residents; patients with primary open-angle glaucoma (POAG); 45 to 89 old at the time of inclusion in the study (middle, elderly and senile ages sensu 2012 WHO classification, www.who.int/ru); clinical refraction in the range of ±6.0 D and astigmatism of ±1.5 D; index of the central corneal thickness (CCT) values from 520 to 580 μ. Hypotensive therapy: a fixed combination of bimatoprost with timolol (FCBT) in conjunction with preservative-free CAI: Dorzolan Solo (Solopharm, Russia).
The exclusion criteria: any other form of primary glaucoma than POAG; change of hypotensive therapy during the observation period; opacities of the optical media preventing the performance of perimetric studies; other retinal diseases (any form of age-related macular degeneration, conditions after occlusions, diabetic retinopathy and its complications, as is customary according to the methodology of clinical trials, https://clinicaltrials.gov); surgical ophthalmological treatment in anamnesis; injuries and diseases of the eye and accessory visual structures; diabetes mellitus and other common diseases requiring hormone therapy.
Among examined patients, 15 (71.5%) were women and 6 (28.5%) were men. The mean age of all patients regardless of their gender at the time of enrollment in the study was 72±8.3; 72.0 (68.2; 78.7) years; for men, the numbers were 65.25±10.4; 65.5 (61.6; 72.8) years; for women, this parameter values were 74.8±5.3; 76.3 (70.0;79.2) years (W=272, p=0.005). POAG was confirmed for 21 (51.2%) eyes, included in Group 1 (observation group). The patients in this group in terms of their age characteristics were as follows: 72.2±9.1; 73.0 (68.4; 78.7) years. Group 2 (control) included patients with suspected glaucoma and ocular hypertension who did not receive any hypotensive therapy: 20 (49.8%) eyes, aged 71.5±6.6; 70.0 (67.6; 78.7) years (W=156, p=0.471). Clinical and demographic data by groups are presented in Table 1.
Table 1. Clinical and demographic characteristics of groups
|
|
Group 1 (n=21) М±σ; Me (Q25%; Q75%) |
Group 2 (n=20) М±σ; Me (Q25%; Q75%) |
Statistical significance |
|
Age (years) |
72.2±9.1; 73.0 (68.4; 78.7) |
71.5±6.6; 70.0 (67.6; 78.7) |
W=156; p=0.471 |
|
Anamnesis (years) |
0.9±0.23; (0.8; 1.0) |
0.73±0.18; 0.7 (0.6; 0.9) |
W=107.5; p=0.006 |
|
Male eyes |
7 |
5 |
X2= 0.058978; p=0.808 |
|
Eye (OD/OS) |
11/10 |
11/9 |
X2=0; p=1.0 |
|
IOL (yes/no) |
14/7 |
10/10 |
X2=0.5862; p=0.443 |
|
PES (yes/no) |
20/1 |
20/0 |
X2=1.2e-32; p=1.0 |
|
SE (D) |
0.48±3.0; 1.8 (-1.8; +2.6) |
-0.26±4.4; 0.9 (-0.5; +2.2) |
W=193; p=0.666 |
|
BCVA |
0.78±0.18; 0.8 (0.7; 0.9) |
0.73±0.18; 0.8 (0.6; 0.9) |
W=180; p=0.432 |
|
AL (mm) |
23.7±0.94; 23.9 (23.6; 24.2) |
23.4±1.2; 22.7 (22.69; 24.5) |
W=59.5; p=0.351 |
|
CCT (μm) |
550±24; 542 (533; 560) |
563±31; 564 (543; 580) |
W=269.5; p=0.123 |
|
MD (dB) |
-5.5±6.4; -2.8 (-6.7; -1.4) |
-2.4±4.3; -0.8 (-2.6; -0.5) |
W=304; p=0.014 |
|
PSD (dB) |
4.7±2.0; 4.5 (2.8; 6.7) |
3.3±1.7; 2.6 (2.2; 3.7) |
W=115; p=0.013 |
|
OD (mm2) |
1.9±0.3; 1.9 (1.7; 2.0) |
2.1±0.5; 1.9 (1.6; 2.5) |
W=225.5; p=0.695 |
|
RNFL (μm) |
74.6±18.7; 78.0 (64.0; 86.0) |
87.7±23.2; 94.5 (68.5; 104.2) |
W=287.5; p=0.044 |
As expected a priori, the groups differed statistically significantly in their structural and functional characteristics, and duration of observation. There were no statistically significant differences in all other clinical and demographic parameters. The magnitude of intraocular pressure (Po) at the time of inclusion in the study was 15.2±4.4; 15.0 (13.0; 18.0) mm Hg in Group 1 vs. 15.25±3.8; 15.0 (13.75; 18.0) mm Hg in Group 2 (p>0.05) (Table 2). Herewith, elasticity increase, characterizing the biomechanical properties of the eyeball fibrous tunic, was in both groups within average values [27-29] (Table 2).
Table 2. IOP level at the onset of the study, and after two and four weeks (mm Hg)
|
|
Measurement method |
Onset of the study* |
4 weeks** |
Statistical significance: * vs. ** |
|
Group 1 |
Ро (iCare) |
15.2±4.4; 15.0 (13.0; 18.0) |
14.6±3.1; 15.0 (13.0; 17.0) |
V=268, p=0.139 |
|
P5 g |
19.3±3.3; 19.5 (16.5; 21.0) |
19.1±3.1; 18.5 (17.0; 21.0) |
V=334.5, p=0.531 |
|
|
P10 g |
24.3±3.9; 24.5 (22.0; 26.0) |
23.6±2.5; 24.0 (21.5; 25.0) |
V=434.5, p=0.212 |
|
|
P15 g |
29.7±4.0; 29.5 (28.0; 31.0) |
29.6±3.2; 29.5 (27.5; 31.5) |
V=327, p=0.716 |
|
|
Elasticity increase |
10.4±2.1; 10.5 (9.5; 12.0) |
10.5±2.8; 10.5 (9.5; 12.0) |
V=249.5, p=0.285 |
|
|
Group 2 |
Ро (iCare) |
15.25± 3.8; 15.0 (13.75; 18.0) |
15.7±1.9; 16.5 (14.5; 17.0) |
V=14.5, p=0.672 |
|
P5 g |
19.2±3.7; 19.3 (16.1; 21.0) |
18.4±2.2; 18.3 (16.4; 20.5) |
V=41, p=0.501 |
|
|
P10 g |
23.6±5.1; 24.3 (18.5; 25.0) |
22.9±2.2; 24.0 (21.3; 25.0) |
V=28, p=1 |
|
|
P15 g |
28.9±5.3; 29.7 (24.8; 30.6) |
28.3±3.0; 27.8 (26.0; 31.1) |
V=41, p=0.905 |
|
|
Elasticity increase |
9.7±2.8; 9.5 (8.4; 10.7) |
9.9±1.5; 10.0 (9.4; 10.8) |
V=25.5, p=0.531 |
|
|
Statistical significance: 1 vs. 2 |
Wi=234.5, pi=0.814 W5=257.5, p5=0.814 W10=279, p10=0.488 W15=265, p15=0.693 Wэ=306, pэ=0.204 |
Wi=183, pi=0.180 W5=270.5, p5=0.609 W10=275, p10=0.540 W15=300, p15=0.254 Wэ=301.5, pэ=0.238 |
|
|
Statistical data processing. Data processing was performed by the R Core Team (2016) (R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria. Source: https://www.R-project.org/). The normality of the data distribution was checked using the Shapiro-Wilk test, while homoscedasticity was examined by the Bartlett’s test. Normally distributed parameters are presented in the format: M±σ, where M is the mean and σ is the standard deviation of the mean. Parameters with a distribution other than normal are presented in the format: Me (Q25%; Q75%), where Me is the median, Q25% and Q75% are quartiles. For comparisons of independent groups or repeated intragroup changes with a normal distribution of parameters, the Student’s t-test was employed; with a non-normal distribution of parameters, the Wilcoxon test was used for comparing several samples; 95% confidence intervals were calculated as M±1.96σ. The critical level of significance for testing statistical hypotheses was assumed at p <0.05.
Results
When replacing the preservative-free CAI as part of the preservative-free combination therapy with FCBT and CAI with Dorzolan Solo (Solopharm, Russia), containing 0.18% sodium hyaluronate and sodium benzoate, after four weeks of treatment we observed no additional statistically significant change in intraocular pressure (p>0.05) (Table 2).
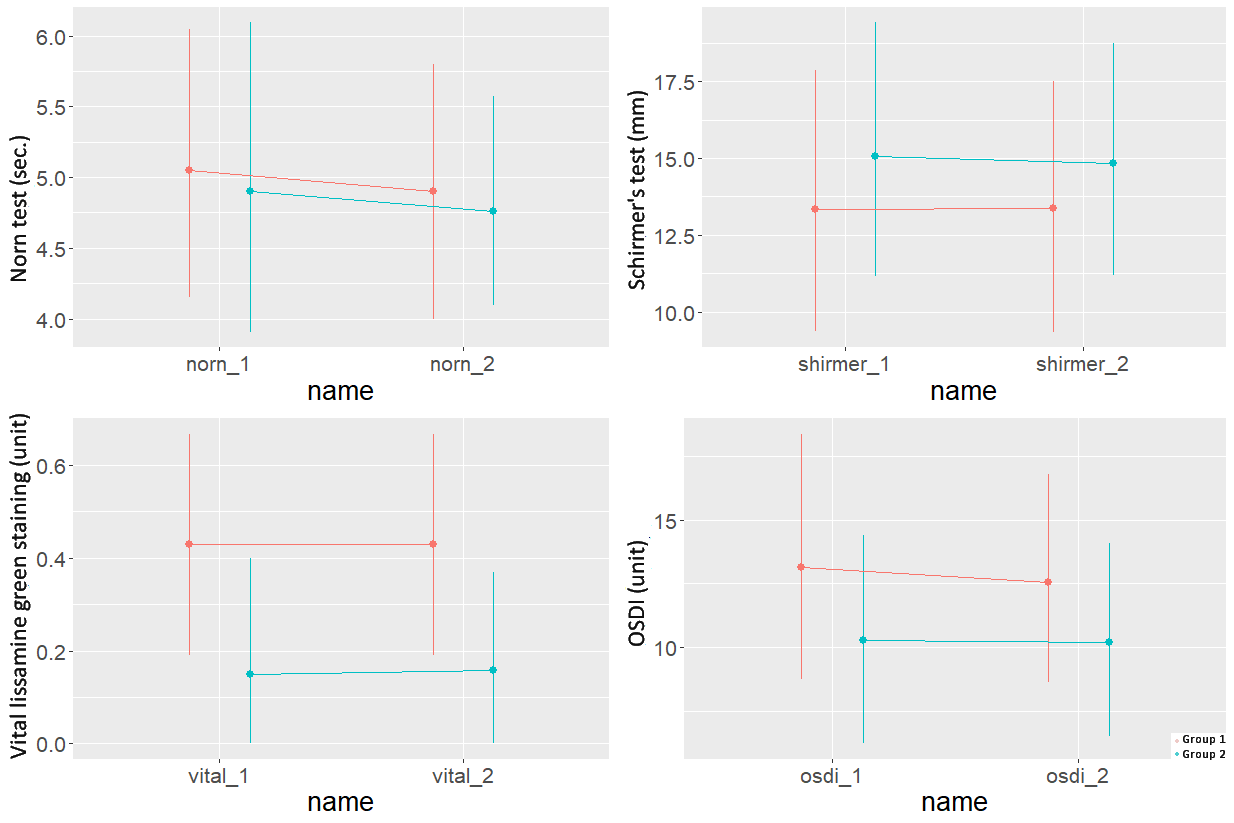
The ocular surface condition sensu TBUT, Schirmer test, vital staining of the ocular surface epithelium with lissamine green, and the OSDI questionnaire at the onset of the study did not differ statistically significant between the groups (Figure 1). During a month of follow-up, after replacing preservative-free Dorzolan Solo (Solopharm, Russia) containing 0.18% sodium hyaluronate with Dorzolan Solo (Solopharm, Russia) containing sodium benzoate as a preservative, all Group 1 patients noted short-term burning during the instillation. To examine the acidity of both preparation options, we used the pH meter on 12 samples (Liquid analyzer Expert-001, Econix-Expert, Russia), three samples for each bottle, two bottles of preservative-free Dorzolan Solo and Dorzolan Solo containing sodium benzoate preservative. The pH was 5.67±0.035 and 5.61±0.04, respectively (p=0.469), which generally corresponds to the declared acidity of the preparation [30].
Figure 1. 95% confidence intervals for the Norn test, Schirmer's test, vital lissamine green staining, OSDI questionnaire at the start of treatment and after 4 weeks.
Herewith, the indicators characterizing eye surface condition in the observation group did not change statistically significantly over the entire observation period. It is worth noting that over a month of observation, Group 2 did not exhibit statistically significant changes in the eye surface as well (Figure 1).
Discussion
To reduce the toxic effects of conventional preservatives on the ocular surface, alternative preservatives have been proposed, such as Purite® oxidizing preservative (Allergan Inc., Irvine, California, USA), Polyquaternium-1 or Polyquad® detergent (Alcon Research, Fort Worth, Texas, USA), SofZia® ion buffer preservative (Novartis AG, Basel, Switzerland), sodium perborate (GenAqua®), and sodium benzoate.
Sodium benzoate is considered a relatively safe preservative [31]. Hypotensive medicines containing sodium benzoate as a preservative do not reduce cell viability and do not change the histological integrity of corneal structures after prolonged exposure. However, they can cause a slight disorder of a barrier function [31]. Consequently, sodium benzoate is considered by some authors as a safer preservative for ophthalmic drugs than BAC [32].
Other existing preservatives developed to reduce toxic effects of BAC are also not completely devoid of this effect. For instance, Polyquad (Polyquaternium) represents a family of polycationic polymers. The name Polyquad refers to about 37 different polymers, but it is Polyquaternium-1 that is most often used in ophthalmology, e.g., as a solution for the treatment of contact lenses [33]. Although Polyquad is considered a relatively safe preservative, some studies have shown that its currently used concentration has a strong toxic effect on the barrier function and viability of corneal cells after the prolonged exposure [32].
Preparations containing SofZia do not have toxicity even after prolonged exposure. Since human cells possess cytochrome oxidase and/or catalase enzymes, SofZia is apparently less toxic to human cells [32, 34-36]; however, it still has some detergent effect [35].
In the course of this study, we examined the effect of a preparation containing sodium benzoate on ocular surface condition. Despite the fact that patients complained of a short-term burning sensation during Dorzolan Solo (Solopharm, Russia) instillation, there was no change in the ocular surface condition over the observation period. As far as we know, this is the first study that investigated the effect of Dorzolan Solo (Solopharm, Russia) containing sodium benzoate on the condition of the ocular surface. In patients with glaucoma, diseases of the eye surface caused by a prolonged use of hypotensive drops are more common, as well as disorders of the tear film due to the filtering bleb. Besides, the presence of concomitant diseases, such as diabetes mellitus, can exacerbate DES [37,38]. Hence, an accurate determination of the potential toxicity of eye drops is of clinical importance in order to avoid unnecessary damage to the eye surface.
Study limitations
There are several limitations to this study. First, the small sample size of the observation group was partially due to the complexity of the patient inclusion criteria, and to some extent, caused by a burning sensation complaint. Second, we used Dorzolan Solo eye drops (Solopharm, Russia) in this study in combination with a preparation of bimatoprost and timolol, and it is impossible to claim that we were able to identify the individual effects of each of these drugs. Finally, it is possible that a month-long observation period was not long enough to reveal a deterioration in the ocular surface condition.
Conclusion
Replacement of the preservative-free CAI in combination therapy of glaucoma with a CAI containing the preservative in the form of sodium benzoate and 0.18% sodium hyaluronate (Dorzolan Solo, Solopharm, Russia) did not yield statistically significant changes in intraocular pressure. At the same time, the eye surface condition did not change statistically significantly during a month of observation. The discomfort caused by the instillation of Dorzolan Solo (Solopharm, Russia), containing sodium benzoate as a preservative, was not related to the pH of the preparation.
Authors’ contribution
All authors have contributed equally.
Financing
No external funding for the project.
Conflict of interest
There is no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
- Ramli N, Supramaniam G, Samsudin A, Juana A, Zahari M, Choo MM. Ocular surface disease in glaucoma: Effect of polypharmacy and preservatives. Optom Vis Sci 2015; 92(9): e222-e226. https://doi.org/10.1097/OPX.0000000000000542.
- Pflugfelder SC, de Paiva CS. The pathophysiology of dry eye disease: What we know and future directions for research. Ophthalmology 2017; 124(11): S4-S13. https://doi.org/10.1016/j.ophtha.2017.07.010.
- Abysheva LD, Avdeev RV, Alexandrov AS, Arapiev MU, Bakunina NA, Baranova NA, et al. Influence of local hypotensive glaucoma therapy on the development and progression of dry eye syndrome. RMJ Clinical ophthalmology 2017; 17(2): 74-82. Russian. https://doi.org/10.21689/2311-7729-2017-17-2-74-82.
- Gomes JAP, Azar DT, Baudouin C, Efron N, Hirayama M, Horwath-Winter J, et al. TFOS DEWS II iatrogenic report. Ocul Surf 2017; 15(3): 511-538. https://doi.org/10.1016/j.jtos.2017.05.004.
- Askeroglu U, Alleyne B, Guyuron B. Pharmaceutical and herbal products that may contribute to dry eyes. Plast Reconstr Surg 2013; 131(1): 159-167. https://doi.org/10.1097/PRS.0b013e318272a00e.
- Koller T., Stürmer J., Gloor B. Risk factors for trabeculectomy failure. Klin Monbl Augenheilkd 1998; 213(07): 1-8. German. https://doi.org/10.1055/s-2008-1034936.
- Movahedan A, Djalilian AR. Cataract surgery in the face of ocular surface disease. Curr Opin Ophthalmol 2012; 23(1): 68-72. https://doi.org/10.1097/ICU.0b013e32834d90b7.
- Baudouin C. Ocular surface and external filtration surgery: Mutual relationships. Dev Ophthalmol 2017; 59: 67-79. https://doi.org/10.1159/000458487.
- Broadway DC, Grierson I, O’Brien C, Hitchings RA. Adverse Effects of topical antiglaucoma medication: II. The outcome of filtration surgery. Arch Ophthalmol 1994; 112(11): 1446-1454. https://doi.org/10.1001/archopht.1994.01090230060021.
- Quigley H, Broman AT. The number of people with glaucoma worldwide in 2010 and 2020. Br J Ophthalmol 2006; 90(3): 262-267. https://doi.org/10.1136/bjo.2005.081224.
- Ali FS, Akpek EK. Glaucoma and dry eye. Ophthalmolog. 2009; 116(6): 1232. https://doi.org/10.1016/j.ophtha.2008.12.034.
- 12. Servat JJ, Bernardino CR. Effects of common topical antiglaucoma medications on the ocular surface, eyelids and periorbital tissue. Drugs Aging 2011; 28(4): 267-282. https://doi.org/10.2165/11588830-000000000-00000.
- Methodologies to diagnose and monitor dry eye disease: Report of the diagnostic methodology subcommittee of the international Dry Eye Workshop (2007). Ocular Surface 2007; 5(2): 108-152. https://doi.org/10.1016/s1542-0124(12)70083-6.
- Fechtner RD, Godfrey DG, Budenz D, Stewart JA, Stewart WC, Jasek MC. Prevalence of ocular surface complaints in patients with glaucoma using topical intraocular pressure-lowering medications. Cornea 2010; 29(6): 618-621. https://doi.org/10.1097/ICO.0b013e3181c325b2.
- Leung EW, Medeiros FA, Weinreb RN. Prevalence of Ocular Surface Disease in Glaucoma Patients. J Glaucoma 2008; 17(5): 350-355. https://doi.org/10.1097/IJG.0b013e31815c5f4f.
- Prum BE, Rosenberg LF, Gedde SJ, Mansberger SL, Stein JD, Moroi SE, et al. Primary Open-Angle Glaucoma Preferred Practice Pattern® guidelines. Ophthalmology 2016; 123(1): 41-111. https://doi.org/10.1016/j.ophtha.2015.10.053.
- Cohen Castel O, Keinan-Boker L, Geyer O, Milman U, Karkabi K. Factors associated with adherence to glaucoma pharmacotherapy in the primary care setting. Fam Pract 2014; 31(4): 453-461. https://doi.org/10.1093/fampra/cmu031.
- Kastelan S., Tomic M., Metez Soldo K., Salopek-Rabatic J. How ocular surface disease impacts the glaucoma treatment outcome. Biomed Res Int 2013; 2013: 696328. https://doi.org/10.1155/2013/696328.
- Nordstrom BL, Friedman DS, Mozaffari E, Quigley HA, Walker AM. Persistence and adherence with topical glaucoma therapy. Am J Ophthalmol 2005; 140(4): 598-606. https://doi.org/10.1016/j.ajo.2005.04.051.
- Dreer LE, Girkin C, Mansberger SL. Determinants of medication adherence to topical glaucoma therapy. J Glaucoma 2012; 21(4): 234-240. https://doi.org/10.1097/IJG.0b013e31821dac86.
- Sleath B, Blalock S, Covert D, Stone JL, Skinner AC, Muir K, et al. The relationship between glaucoma medication adherence, eye drop technique, and visual field defect severity. Ophthalmology 2011; 118(12): 2398-2402. https://doi.org/10.1016/J.OPHTHA.2011.05.013.
- Anwar Z, Wellik SR, Galora A. Glaucoma therapy and ocular surface disease: Current literature and recommendations. Curr Opin Ophthalmol 2013; 24(2): 136-143. https://doi.org/10.1097/ICU.0b013e32835c8aba.
- Freeman PD, Kahook MY. Preservatives in topical ophthalmic medications: Historical and clinical perspectives. Expert Rev Ophthalmol 2009; 4(1): 59-64. https://doi.org/10.1586/17469899.4.1.59.
- Aguayo Bonniard A, Yeung JY, Chan CC, Birt CM. Ocular surface toxicity from glaucoma topical medications and associated preservatives such as benzalkonium chloride (BAC). Expert Opin Drug Metab Toxicol 2016; 12(11): 1279-1289. https://doi.org/10.1080/17425255.2016.1209481.
- Actis AG, Rolle T. Ocular surface alterations and topical antiglaucomatous therapy: a review. Open Ophthalmol J 2014; 8: 67-72. https://doi.org/10.2174/1874364101408010067.
- Baudouin C, Labbé A, Liang H, Pauly A, Brignole-Baudouin F. Preservatives in eyedrops: The good, the bad and the ugly. Prog Retin Eye Res 2010; 29(4): 312-334. https://doi.org/10.1016/j.preteyeres.2010.03.001.
- Avetisov SE, Bubnova IA, Antonov AA. Once more about the diagnostic capacities of elastic tonometry. Vestn Oftalmol 2008; 124(5): 19-22. Russian. https://pubmed.ncbi.nlm.nih.gov/19062552.
- Avetisov SE, Novikov IA, Bubnova IA, Antonov AA, Siplivyi VI. Investigation of the biomechanical properties of the cornea by bidirectional applanation: new approaches to interpreting the results. Vestn Oftalmol 2008; 124(5): 22-25. Russian. https://www.elibrary.ru/item.asp?id=11632986.
- Avetisov SE, Bubnova IA, Petrov SYu, Antonov AA, Reshchikova VS. Peculiarities of biomechanical properties of the fibrous capsule of the eye in patients with primary open-angle glaucoma. National Journal glaucoma 2012; (4): 7-11. Russian. https://www.elibrary.ru/item.asp?id=21063908.
- Loftsson T, Jansook P, Stefánsson E. Topical drug delivery to the eye: Dorzolamide. Acta Ophthalmol 2012; 90(7): 603-608. https://doi.org/10.1111/j.1755-3768.2011.02299.x.
- Nair B. Final report on the safety assessment of benzyl alcohol, benzoic acid, and sodium benzoate. Int J Toxicol 2001; 20(3): 23-50. https://doi:10.1080/10915810152630729.
- Nakagawa S, Usui T, Yokoo S, Omichi S, Kimakura M, Mori Y, et al. Toxicity evaluation of antiglaucoma drugs using stratified human cultivated corneal epithelial sheets. Investigative Ophthalmol Vis Sci 2012; 53(9): 5154-5160. https://doi:10.1167/iovs.12-9685.
- 33. Lipener C; Contact Lens Advisory in Scientific Studies (CLASS) group. A randomized clinical comparison of OPTI-FREE EXPRESS and ReNu MultiPLUS multipurpose lens care solutions. Adv Ther 2009; 26(4): 435-446. https://doi:10.1007/s12325-009-0023-8.
- Kaur IP, Lal S, Rana C, Kakkar S, Singh H. Ocular preservatives: Associated risks and newer. Cutan Ocul Toxicol 2009; 28(3): 93-103. https://doi:10.1080/15569520902995834.
- Kahook MY, Noecker RJ. Comparison of corneal and conjunctival changes after dosing of travoprost preserved with sofZia, latanoprost with 0.02% benzalkonium chloride, and preservative-free artificial tears. Cornea 2008; 27(3): 339-343. https://doi:10.1097/ICO.0b013e31815cf651.
- Baudouin C, Riancho L, Warnet JM, Brignole F. In vitro studies of antiglaucomatous prostaglandin analogues: Travoprost with and without benzalkonium chloride and preserved latanoprost. Investig Ophthalmol Vis Sci 2007; 48(9): 4123-4128. https://doi:10.1167/iovs.07-0266.
- Dorofeev DA, Kryzhanovskaya AV, Tsyganov AZ. Effectiveness of antihypertensive preservative-free therapy with a fixed combination of bimatoprost and timolol (intermediate results). Vestnik Oftalmologii 2020; 136(2): 73-80. Russian. https://doi:10.17116/oftalma202013602173.
- Dorofeev DA, Tur EV, Vizgalova LO, Tryapitsin ID, Ciganov AZ. The effect of preservative-free treatment of primary open-angle glaucoma on the state of ocular surface in combination therapy (intermediate results). Vestnik Oftalmologii 2019; 135(6): 52-59. Russian. https://doi:10.17116/oftalma201913506152.
Received 27 July 2021, Revised 11 March 2022, Accepted 7 June 2022
© 2021, Russian Open Medical Journal
Correspondence to Dmitry A. Dorofeev. Address: 200 Rossiyskaya St., Chelyabinsk 454090, Russia. Phone: +79124778927. E-mail: dimmm.83@gmail.com.